

PROIN ER is the only FDA-approved once-daily medication for the control of canine urinary incontinence (UI) due too urethral sphincter hypotonus.
Indication
PROIN ER is indicated for the control of urinary incontinence due to urethral sphincter hypotonus in dogs.
Benefits
A First-Line UI Treatment Per ACVIM Consensus1
PROIN ER is indicated for the control of canine UI due to urethral sphincter hypotonus.
Treatment Compliance
Simple once-daily dosing helps clients adhere to treatment, which can improve patient outcomes.1
Cost-Effective
Lower cost of treatment can make quality care more accessible and offer long-term affordability for pet owners.
Consistent Control
Patented extended-release technology provides controlled delivery that ensures steady absorption, reducing the occurrence of UI episodes.
Broad Tolerability
Formulated with hydrolyzed protein, making it an alternative for dogs with dietary sensitivities.*
Precautions
PROIN ER may cause hypertension; therefore, use with caution in dogs with pre-existing heart disease, hypertension, liver disease, kidney insufficiency, diabetes, glaucoma, and conditions with a predilection for hypertension.
Use caution in dogs receiving sympathomimetic drugs, tricyclic antidepressants, or monoamine oxidase inhibitors as increased toxicity may result. Use with caution in dogs administered halogenated gaseous anesthetics as this may increase the risk of cardiac arrhythmias.
About PROIN ER
What is PROIN ER?
PROIN ER is a once-daily medication for UI that provides a controlled release of phenylpropanolamine through patented extended-release technology.
The active ingredient in PROIN ER is phenylpropanolamine, which is a chemical analogue of the endogenous sympathomimetic amines. It is an alpha-adrenergic agent which has been reported to increase urethral tone in dogs.3 Its mechanism of action is not well determined, but it is believed to cause the release of norepinephrine by indirectly stimulating the alpha adrenergic receptors of the smooth muscle to increase smooth muscle tone of the urethra, bladder neck, and the internal urethral sphincter.4,5
Mode of Action
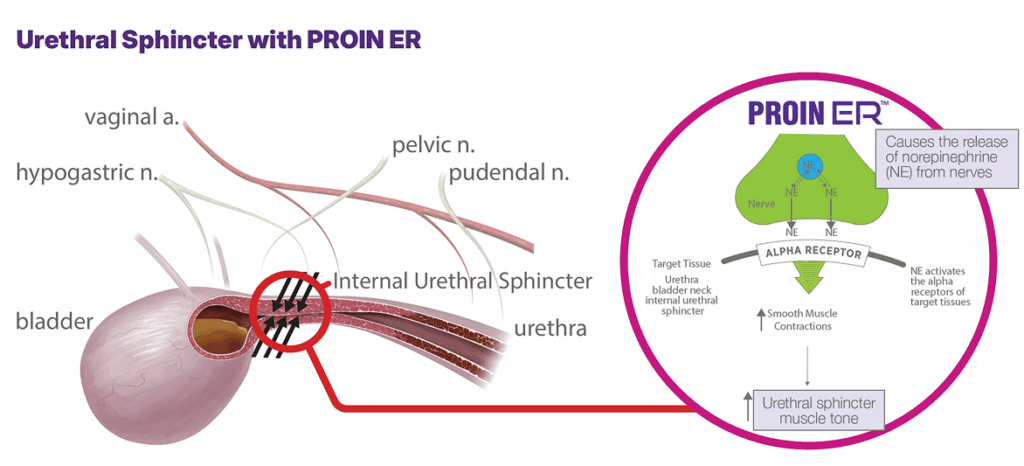
Efficacy & Safety
PROIN ER clinical study
In a multi-centered clinical field study, PROIN ER was administered to 104 client-owned dogs with UI, whose UI was previously controlled with Proin®, for 28 days.6
The study demonstrated that treatment with PROIN ER administered orally once a day was safe and effective for the control of UI due to urethral sphincter hypotonus.
Key Findings:
Consistent Response
99.1% of owners observed maintained or improved continence control within 28 days of PROIN ER administration.
High Acceptance From Pet
82.2% of patients voluntarily consumed a dose of PROIN ER when given with or without a small amount of food.
Maintained UI Control
The level of control (incidence rate) of UI did not differ when animals were transitioned from PROIN® Chewable Tablets to PROIN ER.9
Dosing and Pricing
body weight, once daily. Utilizing the simple weight-band dosing, patient’s should be dosed one PROIN ER tablet* once daily at the tablet strength that corresponds to their body weight. Administer PROIN ER with food. Do not split or crush tablets.
*Dogs exceeding 125 lbs. should receive the appropriate combination of tablets to achieve the recommended dosage.




PROIN ER is available in four different strengths for accurate dosage of virtually any sized dog weighing 10 pounds or more. Plus, the lower cost of treatment can make quality care more accessible and offer long-term affordability for pet owners.
Dosing/mg 1x per day
9 – 36 mg
Days
30
As Low As
$0.76/day
PROIN ER SKU mg Size
18 mg for dogs 10-20 lbs.
Dosing/mg 1x per day
18.9 – 72 mg
Days
30
As Low As
$1.00/day
PROIN ER SKU mg Size
38 mg for dogs 21-40 lbs.
Dosing/mg 1x per day
36.9 – 144 mg
Days
30
As Low As
$1.36/day
PROIN ER SKU mg Size
74 mg for dogs 41-80 lbs.
Dosing/mg 1x per day
72.9 – 225 mg
Days
30
As Low As
$1.66/day
PROIN ER SKU mg Size
145 mg for dogs 81-125 lbs.
Treatment Considerations
Due to the active ingredient in PROIN ER, phenylpropanolamine, it is recommended to perform a baseline systolic blood pressure before starting PROIN ER since hypertension is a potential side effect of alpha agonist therapy. Dogs treated with PROIN ER should also have their blood pressure monitored after 2-4 weeks on the medication. Other adverse effects of phenylpropanolamine include restlessness, aggression, decreased appetite, and insomnia.
About Canine UI
Urethral Sphincter Mechanism Incompetence
Urethral sphincter mechanism incompetence (USMI) is the most common cause of urinary incontinence in dogs affecting up to 20% of neutered female dogs and 30% of those over 44 lbs. It is less commonly reported in neutered males and intact dogs of both sexes. In most dogs it occurs within 3 years of neutering, although in many it may not become a major problem until later in life when it can be complicated by diseases that cause polyuria and polydipsia.1,10
Canine UI Patient Profile
Predisposed Breeds
Large dogs are more likely to develop UI due to urethral sphincter hypotonus. Among the most common breeds affected are the Old English sheepdog, Doberman pinscher, Boxer, German Shepherd Dog, and Weimeraner.10 A recent study indicates that earlier spaying/neutering may increase the risk of development of urinary sphincter mechanism incompetence in dogs with a projected adult body weight greater than 33 lbs.10
The Impact of Canine UI
Inappropriate urination due to urethral sphincter hypotonus most commonly happens when a dog is sleeping or relaxing, meaning that the comfy places they love most (couches, beds, carpets) become off limits.
74% of dog owners indicate “leaking” would impact sharing space with their dog.2
68% of dog owners indicate “leaking” would affect decisions about whether to include their dog in personal activities.2
Resources
PROIN ER Detailer
Educate your team about canine UI and PROIN ER.
PROIN ER Pet Owner Brochure
Teach your clients about UI and how PROIN ER can help.
References
1Kendall A, Byron JK, Westropp JL, Coates JR, Vaden S, Adin C, Oetelaar G, Bartges JW, Foster JD, Adams LG, Olby N, Berent A. ACVIM consensus statement on diagnosis and management of
urinary incontinence in dogs. J Vet Intern Med. 2024 Mar-Apr;38(2):878-903. doi: 10.1111/jvim.16975. Epub 2024 Jan 13. PMID: 38217372; PMCID: PMC10937496.
2Barter LS et al. 2015; Adams VJ et al. J Small Anim Pract. 2005.
3Richter K.P., Ling G.V. Clinical response and urethral pressure profile changes after phenylpropanolamine in dogs with primary sphincter incompetence. JAVMA, Vol. 187, No 6, September 15, 1985. 605-611.
4Scott, L., Leddy M. and Bernay, F. Evaluation of phenylpropanolamine in the treatment of urethral sphincter mechanism incompetence in the bitch. J. Small Anim. Pract. 2002;43(11): 493-6.
5Noel, S., et al. Combined pharmacokinetic and urodynamic study of the effects of oral administration of phenylpropanolamine in female Beagle dogs. Vet. Journal, 2010; 184(2): 201-207.
6Pegasus Laboratories, Inc. (2019). Freedom of Information (FOI) Summary for PROIN ER (phenylpropanolamine hydrochloride extended-release tablets) Dogs: U.S., (NADA 141-517). Center
for Veterinary Medicine (CVM).
7Kynetec 2022 Urinary Incontinence in Canine Survey.
8Consumer Dog Owner survey conducted by PRN Pharmacal, December 2022.
9FDA-CVM FOI Summary, PROIN ER, NADA 141-517 (March 29, 2019)
10Byron JK, Taylor KH, Phillips GS, Stahl MS. Urethral Sphincter Mechanism Incompetence in 163 Neutered Female Dogs: Diagnosis, Treatment, and Relationship of Weight and Age at Neuter to Development of Disease. J Vet Intern Med 2017;31:442–448.
*While hydrolyzed proteins reduce the risk of immunologic reactivity, individual sensitivities may still occur.